Authors
Léa Mazot, Sandra Robrecht, Adam Giza, Anna-Maria Fink, Julia v. Tresckow, Petra Langerbeins, Florian Simon, Moritz Fürstenau, Othman Al-Sawaf, Kirsten Fischer, Eugen Tausch, Ludwig Fischer von Weikersthal, Clemens-Martin Wendtner, Karl-Anton Kreuzer, Sebastian Böttcher, Matthias Ritgen, Stephan Stilgenbauer, Barbara Eichhorst, Michael Hallek, Paula Cramer.
Introduction
While the armamentarium for the treatment of chronic lymphatic leukemia (CLL) has increased and improved, the best treatment sequence remains unclear. Specifically, there is a debate whether patients who relapse after time-limited venetoclax-based therapy should undergo re-treatment or switch to a different mechanism of action, namely BTK inhibition. The CLL2-BAG trial is one of the first phase-II trials which evaluated the now well-established combination of Venetoclax-Obinutuzumab (Ven-Obi) with an individualized measurable residual disease (MRD) guided treatment duration in an all-comer population irrespective of prior treatments, fitness and CLL risk factors (Cramer et al, Lancet Oncol. 2018). During the conduct of the trial, the option of a re-treatment with Ven-Obi was included in the trial and offered to patients with a progression in need of treatment. This is the first analysis of all seven patients with two consecutive treatment lines with Ven-Obi in phase-II CLL2-BAG trial.
Methods
Between May 2015, and Jan 2016, a total of 66 patients were enrolled in the CLL2-BAG trial and received Ven-Obi in an induction phase (8 cycles with 28 days each) and maintenance phase (up to 8 cycles with 84 days) depending on individual response and MRD in peripheral blood (PB). Two option-al cycles of bendamustine debulking could be administered in patients with high tumor load before Ven-Obi. In case of progression with need of treatment as per iwCLL criteria ≥6 months after end of Ven-Obi treatment, a re-treatment with Ven-Obi could be administered in the clinical trial.
Results
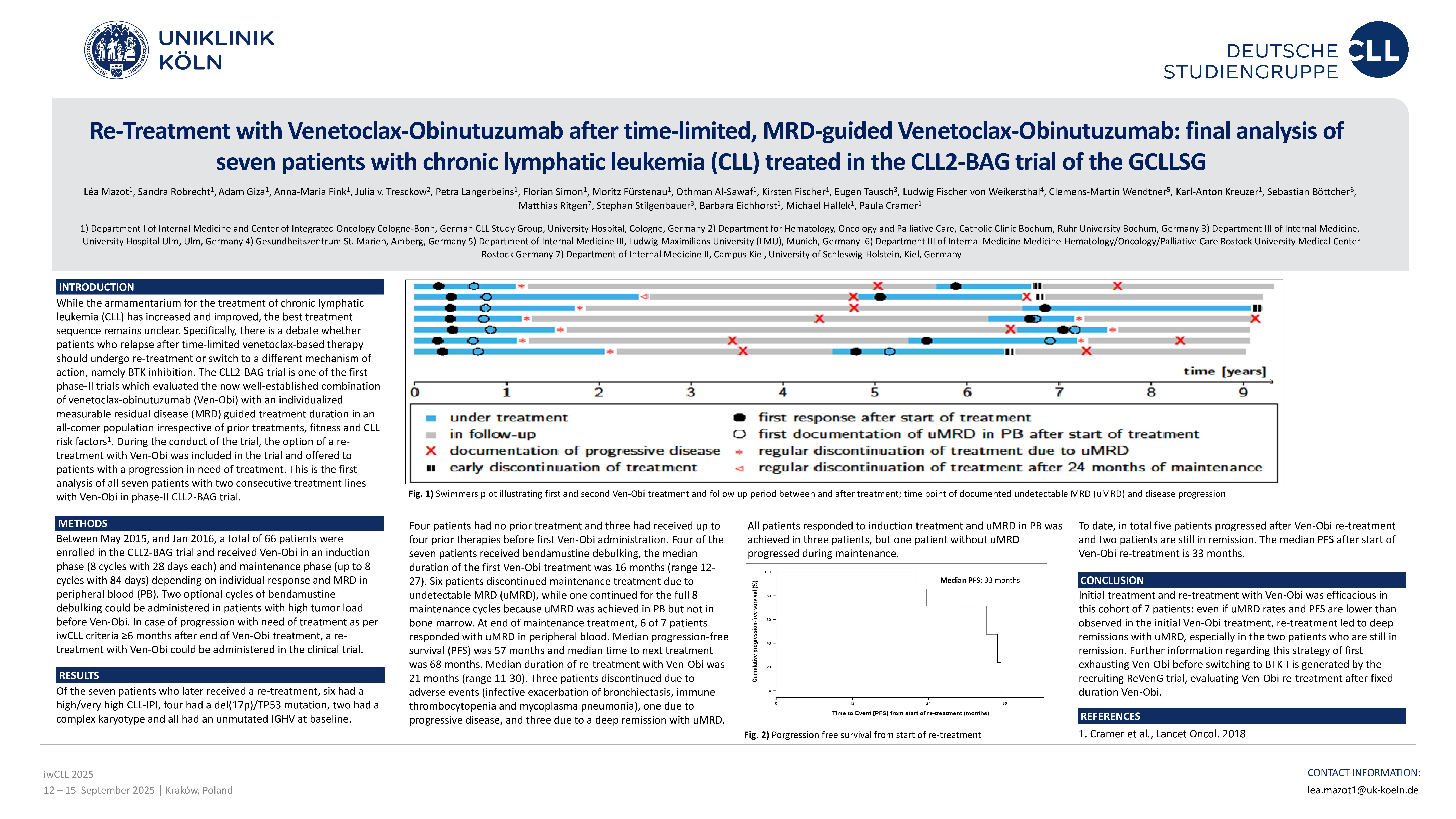
Of the seven patients who later received a re-treatment, six had a high/very high CLL-IPI, four had a del(17p)/TP53 mutation, two had a complex karyotype and all had an unmutated IGHV at base-line. Four patients had no prior treatment and three had received up to four prior therapies before first Ven-Obi administration. Four of the seven patients received bendamustine debulking, the median dura-tion of the first Ven-Obi treatment was 16 months (range 12-27). Six patients discontinued mainte-nance treatment due to undetectable MRD (uMRD), while one continued for the full 8 maintenance cycles because uMRD was achieved in PB but not in bone marrow. At end of maintenance treatment, 6 of 7 patients responded with uMRD in peripheral blood. Median progression-free survival (PFS) was 57 months and median time to next treatment was 68 months.
Median duration of re-treatment with Ven-Obi was 21 months (range 11-30). Three patients discontin-ued due to adverse events (infective exacerbation of bronchiectasis, immune thrombocytopenia and mycoplasma pneumonia), one due to progressive disease, and three due to a deep remission with uMRD. All patients responded to induction treatment and uMRD in PB was achieved in three patients, but one patient without uMRD progressed during maintenance. To date, in total five patients pro-gressed after Ven-Obi re-treatment and two patients are still in remission. The median PFS after start of Ven-Obi re-treatment is 33 months
Conclusion
Initial treatment and re-treatment with Ven-Obi was efficacious in this cohort of 7 pa-tients:, even if uMRD rates and PFS are lower than observed in the initial Ven-Obi treatment, re-treatment led to deep remissions with uMRD, especially in the two patients who are still in remission. Further information regarding this strategy of first exhausting Ven-Obi before switching to BTK-I is generated by the recruiting ReVenG trial, evaluating Ven-Obi re-treatment after fixed duration Ven-Obi.
Keywords : CLL, Venetoclax, Obinutuzumab
Please indicate how this research was funded. :
Please indicate the name of the funding organization.: