Add a file to start using this block
Authors
Dafne Barozzi, Federica Barbaglio, Daniela Belloni, Davide Ribezzi, Silvia Farè, Riccardo Pinos, Marta Sampietro, Margherita Pauri, Elisa Albi, Francesca Martini, Paolo Ghia and Cristina Scielzo
Introduction
Bioengineering three-dimensional (3D) lymphoid tissues represent a rapidly expanding area in oncohematology, offering strong translational potential for modeling cancer cells-tumor microenvironment (TME) interactions. Due to the challenges in optimizing models that can incorporate the TME and capture the dynamicity of blood cancers, this research field is still poorly explored [Barozzi,D. et al; 2023]. In this study we exploited our lymphoid-like 3D in vitro dynamic model to investigate the physio-pathological mechanisms of Chronic Lymphocytic Leukemia (CLL). The TME plays a central role in CLL progression, relapse and therapy resistance [Caligaris-Cappio,F. et al; 2014], driving the development of targeted therapies to disrupt related key crosstalk pathways. This underscores the need for efficient in vitro models reflecting tissue-specific contexts
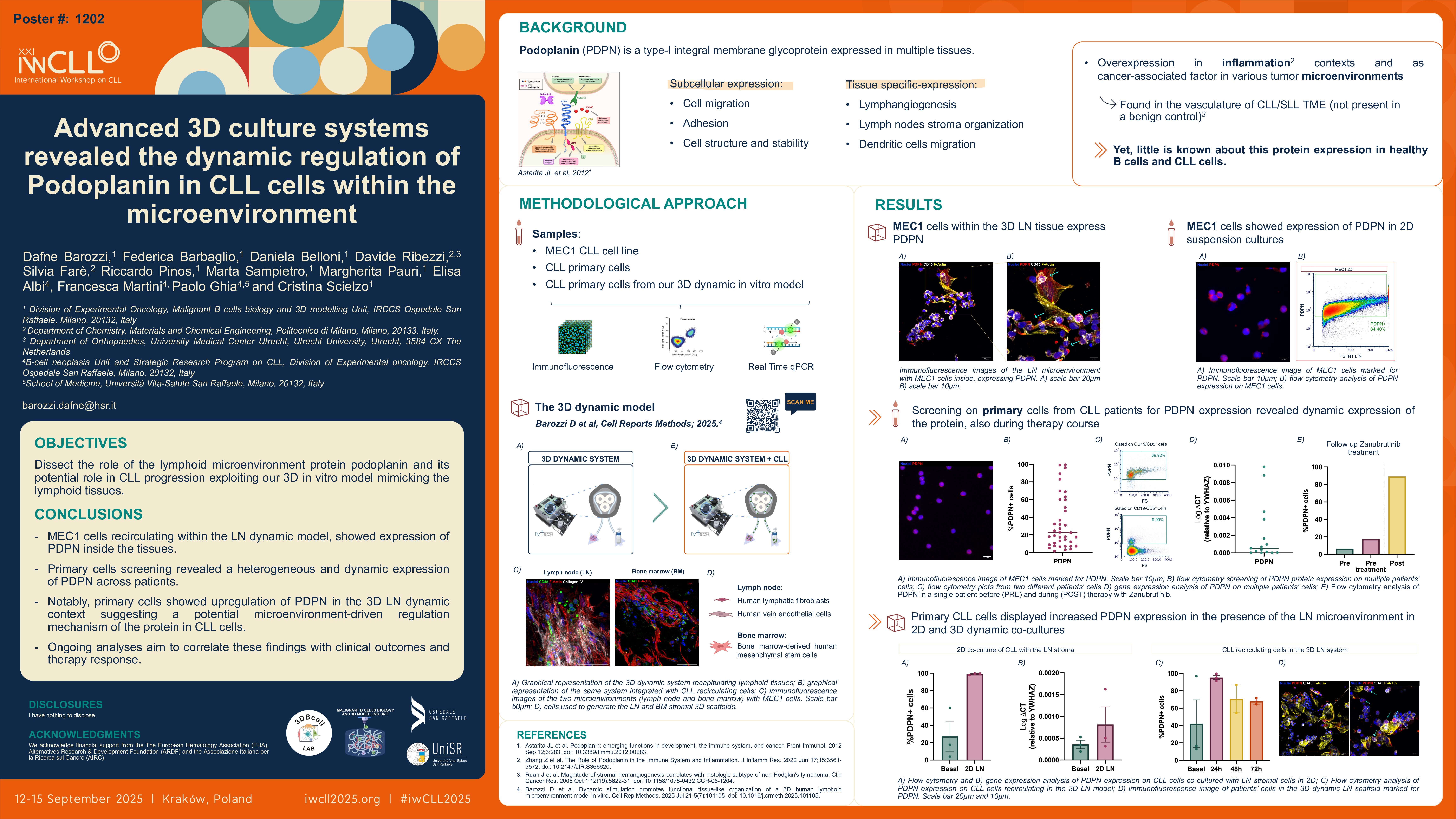
Our system allowed us to reproduce both bone marrow (BM) and lymph node (LN) microenvironments, recreating the journey of CLL cells disseminating through the circulation. We were able to investigate various aspects of the leukemia-TME crosstalk, observing the expression of podoplanin (PDPN) on the neoplastic clone and not only by the stromal cells as expected. PDPN is known in various biological contexts, particularly as central regulator of fibroblastic reticular cells and endothelium within the LN. Interestingly its expression in B lymphocytes and CLL cells is not well-established, yet it is reported its overexpression in inflammation contexts and as cancer‑associated factor in various tumor microenvironments such as in the vasculature of CLL/SLL TME, if compared with the benign control. [Ruan,J. et al; 2006].
Methods
The 3D system consists of a peristaltic pump which allows the circulation of CLL cells inside and outside two different bioreactors containing lymphoid-like 3D constructs. The tissue-like samples were generated seeding human mesenchymal stromal cells (for the BM model) and human lymphatic fibroblasts together with endothelial cells (for the LN model) on a biocompatible porous scaffold. The first was developed inside a bioreactor under dynamic tangential flow at a perfusion rate of 100 μl/min, while the LN underwent a bottom-to-top sigmoidal flow at a prefusion rate of 200 μl/min. Leukemic cells were added to the recirculation after 15 days of culture reflecting the diagnostic threshold for the total leukocyte count: 5×106 cells/ml. Recirculating cells were withdrawn at two different timepoints (24 and 72h after the circulation start point) and key functional markers were analyzed through flow cytometry, gene expression analysis and protein quantification. The 3D scaffolds were processed for confocal imaging and histochemistry analysis, together with gene and protein expression analysis.
Results
We successfully optimized a perfused 3D in vitro model mimicking both BM and LN tissues, demonstrating its ability in sustaining CLL cell viability while replicating critical interactions with the TME [Barozzi,D. et al. accepted for publication]. Using both a CLL cell line (MEC1) and primary patient-derived cells, we confirmed that leukemic cells can infiltrate and populate both 3D BM and LN microenvironments, showing time-dependent changes in key functional markers compared to 2D cultures. In response to biochemical and mechanical stimuli (e.g. shear stress and compression), leukemic cells modulated the expression of key functional markers, involved in homing and dissemination (e.g. CXCR4), adhesion-related proteins involved in retention of CLL cells inside the tissues (e.g. CD44/hyaluronic acid), and in transendothelial migration (e.g. VLA-4/fibronectin and VCAM-1). While assessing spatial distribution and organization of CLL cells inside the 3D constructs, we interestingly noted the expression of PDPN in both MEC1 and primary cells. We are now screening patients’ cells (n=24 so far), observing by flow cytometry a highly heterogeneous inter-patient expression of PDPN in the neoplastic clone – ranging from 4% to 90% of PDPN+ cells – contrarily to a more homogeneous expression observed in healthy B cells from different donors, remaining around 70%. Preliminary data using our 3D dynamic system, revealed a significant increase in PDPN+ CLL cell in circulation in presence of the LN microenvironment but not in the BM, respectively if compared to their basal levels.
Conclusion
We generated a reliable 3D in vitro model to reproduce the CLL-TME crosstalk. In particular, we observed the dynamic expression of PDPN in primary CLL cells from different donors. The regulation of this microenvironment-dependent protein could possibly be associated with disease progression, transformation, or microenvironmental interactions. A deep analysis is ongoing also correlating with the clinical outcome and response to therapies.
Keywords : 3D, microenvironment, perfusion, bioengineering, biomaterial, lymphoid tissues, leukemia
Please indicate how this research was funded. :
Please indicate the name of the funding organization.: