Authors
Xutao Guo, Yunxia Zhang, Xiaolei Wei, Yongqiang Wei, Haohao Lei, Ning Su, Ru Feng.
Background
BTK inhibitors (BTKi) are recommended for long-term therapy in chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) but face challenges including limited depth of response, high dropout rates, and acquired mutations, hindering optimal survival benefits. In fit patients, exploration of time-limited therapy models such as fixed-duration or MRD-guided therapies has shown promising results, highlighting the need for similar investigations in unfit patients.
Methods
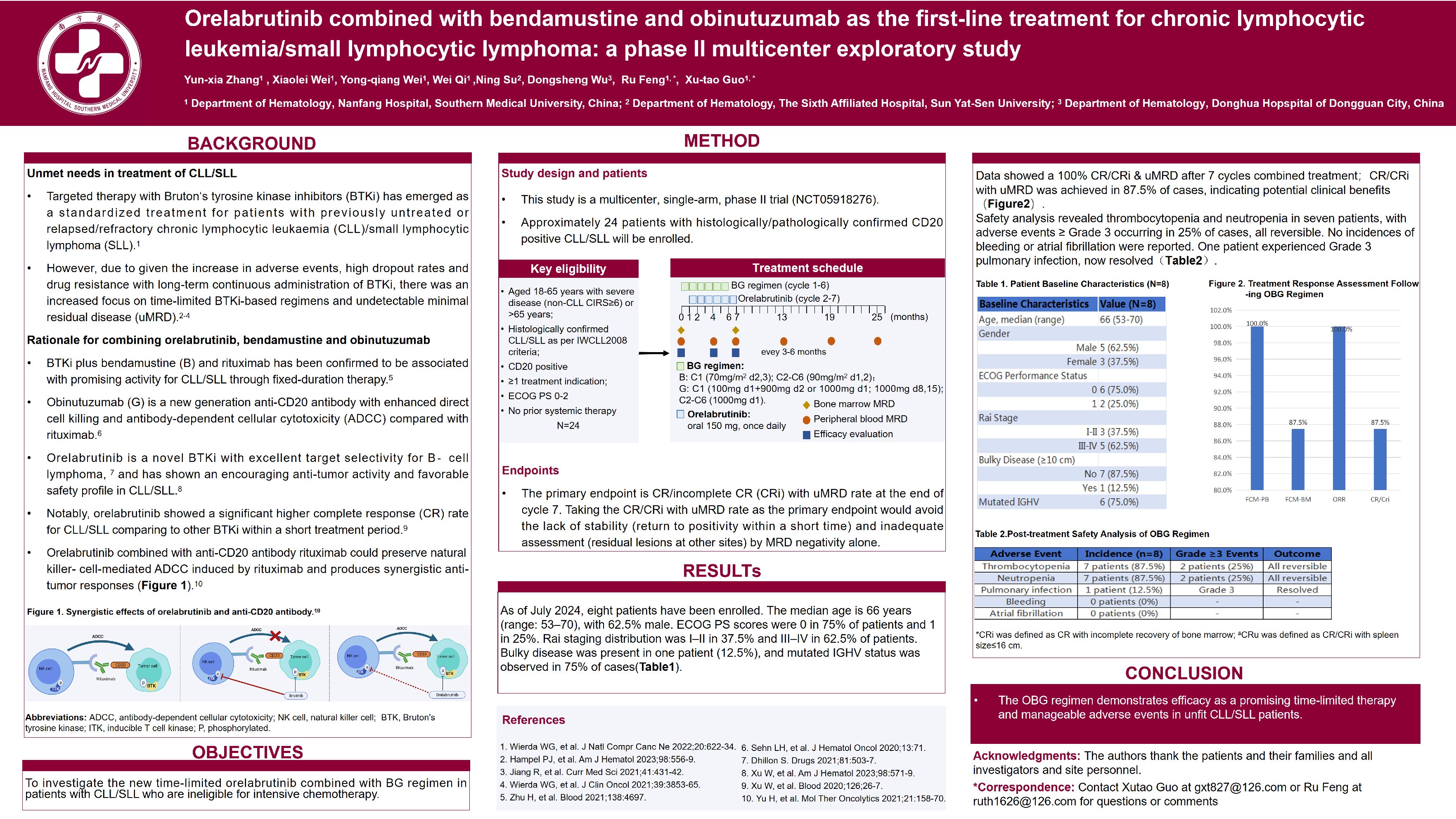
We explored a time-limited therapy model combining orelabrutinib, bendamustine, and obinutuzumab (OBG) in unfit CLL/SLL patients. Orelabrutinib’s superior efficacy and safety profile were chosen based on its selective BTK inhibition and minimal impact on ITK kinase, preserving obinutuzumab’s ADCC effect. Obinutuzumab’s enhanced ADCC and ADCP capabilities were leveraged alongside bendamustine’s cytotoxicity. The primary endpoints include the rate of complete response (CR) combined with undetectable minimal residual disease (uMRD) at the end of 7 cycles (CR/CRi & uMRD, with patients achieving incomplete CR categorized as CRi). Secondary endpoints encompass CR/CRu & uMRD rates at the end of 7 cycles (CRu defined as splenic longest diameter ≤ 16 cm with other criteria meeting CR/CRi standards), uMRD rate (detected via NGS at a level of 10^-6), progression-free survival (PFS), and overall survival (OS).
Results
As of July 2024, eight patients have been enrolled in our study. The data showed a 100% complete response rate after 6 cycles, with peripheral blood and bone marrow uMRD rates of 100% and 87.5%, respectively. CR with uMRD was achieved in 87.5% of cases, indicating potential clinical benefits. Safety analysis revealed that among the eight patients evaluated, thrombocytopenia and neutropenia were observed in seven patients, with adverse events ≥ Grade 3 occurring in 25% of cases, all of which were reversible. No incidences of bleeding or atrial fibrillation were reported. One patient experienced Grade 3 pulmonary infection, which has since resolved.
Conclusion
The OBG regimen demonstrates efficacy as a promising time-limited therapy in unfit CLL/SLL patients, achieving high rates of complete response (CR) combined with undetectable minimal residual disease (uMRD). Safety analysis indicates that the OBG regimen is well-tolerated, with manageable adverse events such as reversible thrombocytopenia and neutropenia observed in a subset of patients. These findings support further investigation of the OBG regimen to optimize treatment outcomes in this patient population.
Keywords : CLL,Orelabrutinib,Obinutuzumab
Please indicate how this research was funded.:
Please indicate the name of the funding organization. :