Authors
F.Autore, A.Visentin, L.Schiattone, J,Olivieri, M.Deodato, D.Giannarelli, C.Vitale, A.Sanna, I.Scortechini, R.Fazzi, M.I.DelPrincipe, N.Maschio, S.LeonettiCrescenzi, S.Vaccarini, P.Sportoletti, A.Gozzetti, I.Innocenti, L.Stirparo, F.Angotzi, G.Ricciuti, L.Carriero, F.Perutelli, R.Pasquale, A.M.Frustaci, A.Tedeschi, L.Trentin, C.Cattaneo, L.Pagano, L.Laurenti.
Infections are a major source of morbidity and mortality in patients with Chronic Lymphocytic Leukemia (CLL). The development of targeted agents decreased this rate but patients, often elderly, with comorbidities, heavily treated, experienced serious infectious complications.
The aim of our study is to describe in a real-world setting the infectious complications in patients with CLL treated with second generation Bruton’s Tyrosine Kinase inhibitors (BTKis) Acalabrutinib (Acala) and Zanubrutinib (Zanu) in routine clinical practice, and to identify additional factors of infectious risk.
This retrospective multicenter analysis was conducted on 460 CLL patients: 324 treated with Acala and 136 with Zanu. Patients of the second group were older, more frequently had TP53mut, pulmonary and renal impairment, more advanced Rai stage and more often already treated.
We recorded 164 events of infections during treatment with Acala, occurring in 110 patients (34%) with an occurrence rate of 0.51. The total number of G3-G4 infections was 47 in 41 patients (12.7%) with an occurrence rate of 0.14.
We recorded 71 events of infections during treatment with Zanu, occurring in 49 patients (36%) with an occurrence rate of 0.52. The total number of G3-G4 infections was 15 in 13 patients (9.6%) with an occurrence rate of 0.11.
We registered a trend toward an earlier median time to infection with Zanu (4.5 months vs 8.2 months, p=0.068) and a significant median time to infections G3-G4 with Zanu (3.1 months vs 7.8 months, p=0.025)
Risk factors for infection all grade in Acala were age at treatment, diabetes, chronic obstructive pulmonary disease (COPD), CrCl < 70, CIRS, previous lines of treatments, infections and/or pneumonia in the previous 12 months; in Zanu were diabetes, Rai stage, infections and/or pneumonia in the previous 12 months.
If considering only G3-G4 infections, COPD and pneumonia in the previous 12 months were the unique significant variable for Acala (p < 0.001), pneumonia in the previous 12 months for Zanu (p < 0.001).
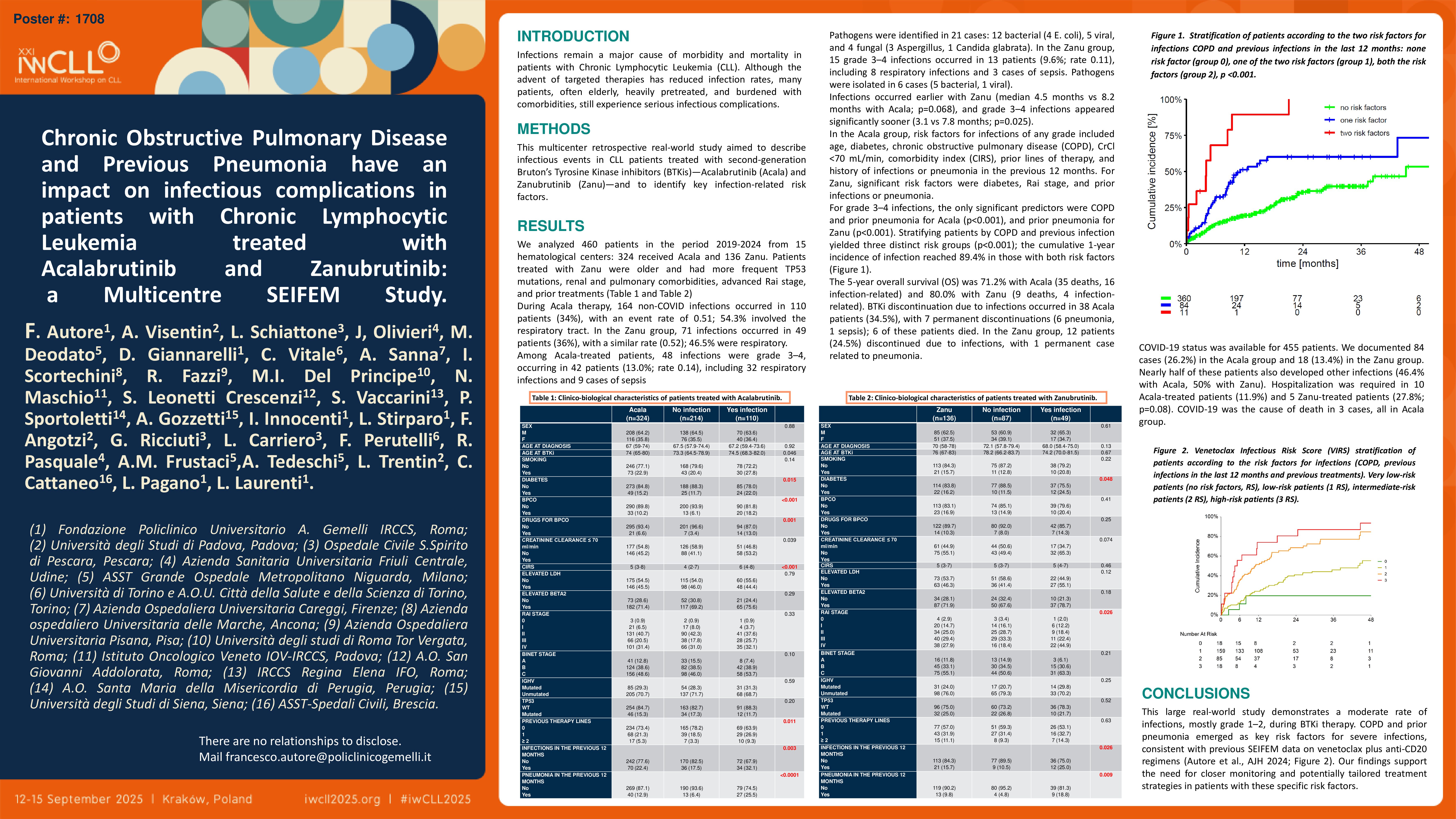
Stratifying patients according to COPD and previous infections in the last 12 months we obtained 3 groups, significantly different in terms of risk for infection (p < 0.001). The cumulative incidence of infection was 89.4% in the first year if patients showed both characteristics.
We recorded a 5-years overall survival OS of 71,2% per Acala with 35 deaths, of whom 16 due to infections. For Zanu a 5-y OS of 80.0% with 9 deaths, of whom 4 due to infections.
Acala was withdrawn for infections in 38 patients (34.5%), 7 of them were definitive: mainly pneumonia (6 cases) and sepsis (1 cases); 6 patients in this subgroup died for these infections in BTKi.
Zanu was withdrawn for infections in 12 patients (24.5%), 1 of them was definitive due to pneumonia.
This large study showed a mild rate of infections, most of grade 1-2. We found risk factors for G3-G4 infections COPD and previous pneumonia, confirming a role of these risk factors already described with venetoclax associated to anti-CD20 antibody in previous analysis from SEIFEM (Autore et al., AJH 2024). According to our analysis, we could consider selecting patients on BTKis therapy with specific risk factors for infection such as COPD and previous pneumonia, to be better and more closely monitored.
Keywords : LLC, acalabrutinib, zanubrutinib, infections, COPD, pneumonia
Please indicate how this research was funded. : none
Please indicate the name of the funding organization.: none