Authors
E. ÇALLIKU, A. PEROLLA, V. SEMANAJ, D. ROKO, T. DEDEJ-KURTI, S. GRAZHDANI, A. BABAMETO, A. IVANAJ
Introduction
It is quite uncommon for a person to have both acute myeloid leukemia (AML) and chronic lymphocytic leukemia (CLL) at the same time, especially when they first show up and haven’t had any cytotoxic medication before. It is even less likely to have genetic problems at the same time, including t(8;16)(p11.2;p13.3) and del(17p), which are both linked to aggressive disease types and bad outcomes. Some studies say that these two cancers may be caused by the same leukemogenic pathways, clonal evolution, or a problem with the same hematopoietic stem cell.
We did a thorough diagnostic workup that included a peripheral blood smear, a bone marrow aspiration, flow cytometry (FCM), cytogenetics, and molecular analysis using FISH. We also did a detailed study of the literature to put the results in context and look at what they might mean for clinical practice.
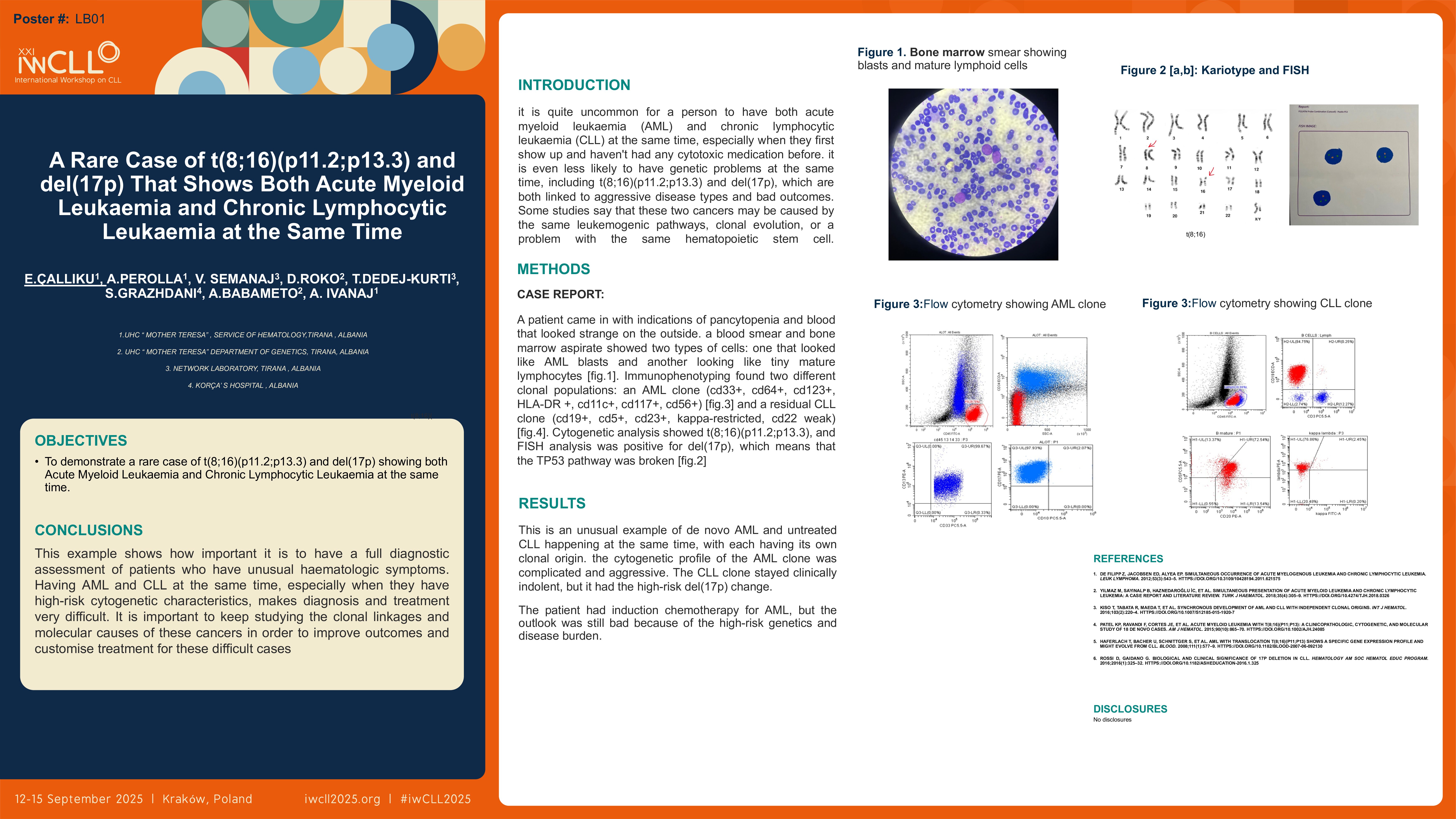
Case Report
A patient came in with indications of pancytopenia and blood that looked strange on the outside. A blood smear and bone marrow aspirate showed two types of cells: one that looked like AML blasts and another that looked like tiny mature lymphocytes. Immunophenotyping found two different clonal populations: an AML clone (CD33+, CD64+, CD123+, HLA-DR+, CD11c+, CD117+, CD56+) and a residual CLL clone (CD19+, CD5+, CD23+, kappa-restricted, CD22 weak). Cytogenetic analysis showed t(8;16)(p11.2;p13.3), and FISH analysis was positive for del(17p), which means that the TP53 pathway was broken.
Results
This is an unusual example of de novo AML and untreated CLL happening at the same time, with each having its own clonal origin. The cytogenetic profile of the AML clone was complicated and aggressive. The CLL clone stayed clinically indolent, but it had the high-risk del(17p) change. The patient had induction chemotherapy for AML, but the outlook was still bad because of the high-risk genetics and disease burden.
Discussion
It’s still not clear how AML and CLL can show up at the same time. Reports by Yılmaz, Kiso, and DeFilipp, as well as other research, imply that genetic instability, clonal haematopoiesis, and shared stem cell origins may be the cause of this. The existence of t(8;16), which is commonly linked to monocytic AML and poor response to treatment, along with del(17p) CLL, shows how complicated biology is and how hard it is to treat. It is important to note that del(17p) is associated to TP53 disruption, which affects both the development of leukaemia and the failure of treatment in CLL. It may also play a role in triggering transformation. It is hard to take care of these kinds of patients, and each one needs their own plan. It may be necessary to use targeted medicines and think about stem cell transplantation.
Conclusions
This example shows how important it is to have a full diagnostic assessment of patients who have unusual haematologic symptoms. Having AML and CLL at the same time, especially when they have high-risk cytogenetic characteristics, makes diagnosis and treatment very difficult. It is important to keep studying the clonal linkages and molecular causes of these cancers in order to improve outcomes and customise treatment for these difficult cases.
Keywords : Concomitant leukemia, Therapy-naïve AML and CLL, Hematologic malignancies, Prognostic cytogenetics
Please indicate how this research was funded. : This work received no external funding.
Please indicate the name of the funding organization.: n/a